Betulinic Acid and Cancer: Nature’s Promising Warrior Against Malignancy
Listen to this information anywhere by hitting play below:
From Nature’s Pharmacy: Betulinic Acid
Cancer remains one of humanity’s most formidable adversaries, claiming millions of lives annually despite decades of intensive research and treatment innovation. While conventional therapies like chemotherapy and radiation have saved countless lives, their significant side effects and the persistent challenge of drug resistance have driven scientists to explore nature’s pharmacy for gentler, more selective alternatives. Enter betulinic acid: a naturally occurring compound that has captured the attention of cancer researchers worldwide for its remarkable ability to selectively destroy cancer cells while leaving healthy tissue largely unharmed (Pisha et al., 1995).
A Serendipitous Discovery in Birch Bark
The story of betulinic acid (BA) as a cancer-fighting agent began in 1995 when researchers at the University of Illinois at Chicago made a groundbreaking discovery. Through bioassay-guided fractionation, they identified this pentacyclic triterpene as a melanoma-specific cytotoxic agent, extracted from the bark of white birch trees (Betula species). What made this discovery particularly exciting was not just BA’s ability to kill melanoma cells, but its selective toxicity—meaning it attacks cancer cells while sparing normal cells, and when tested in athymic mice carrying human melanomas, tumor growth was completely inhibited without any observable toxicity (Pisha et al., 1995; Pezzuto, 1995).
This selectivity is revolutionary. Traditional chemotherapy drugs often cannot distinguish between rapidly dividing cancer cells and healthy cells that also divide frequently, such as those in hair follicles, the gastrointestinal tract, and bone marrow: hence the notorious side effects of hair loss, nausea, and immune suppression. Betulinic acid’s selective nature suggested a fundamentally different mechanism of action, one that could potentially transform cancer treatment (Fulda, 2008; Mullauer et al., 2010).
The Molecular Mechanism: A Direct Strike on Cancer’s Power Plant
What makes betulinic acid so remarkably selective? The answer lies in its unique mechanism of action. Unlike many chemotherapeutic agents that work through various indirect pathways, BA takes a direct approach by targeting the mitochondria—the powerhouse of the cell (Fulda, 2008; Fulda & Kroemer, 2009; Mullauer et al., 2010).
The Mitochondrial Pathway to Death
BA induces apoptosis (programmed cell death) through what researchers call the “intrinsic” or “mitochondrial” pathway. Here’s how it works: BA directly disrupts the mitochondrial membrane potential, causing mitochondrial membrane permeabilization. This leads to the release of cytochrome c from the mitochondria into the cytoplasm, which then triggers a cascade of caspase enzymes—molecular executioners that systematically dismantle the cell (Fulda, 2008; Mullauer et al., 2010; Tan et al., 2003; Wang et al., 2019).
What’s particularly fascinating is that this process appears to be largely independent of p53, a tumor suppressor gene that is frequently mutated in cancers. This means BA can potentially work even in cancers that have developed resistance to therapies that depend on functional p53, offering hope for treating notoriously difficult-to-treat malignancies. This is a huge beneficial aspect (Mullauer et al., 2010; Selzer et al., 2000).
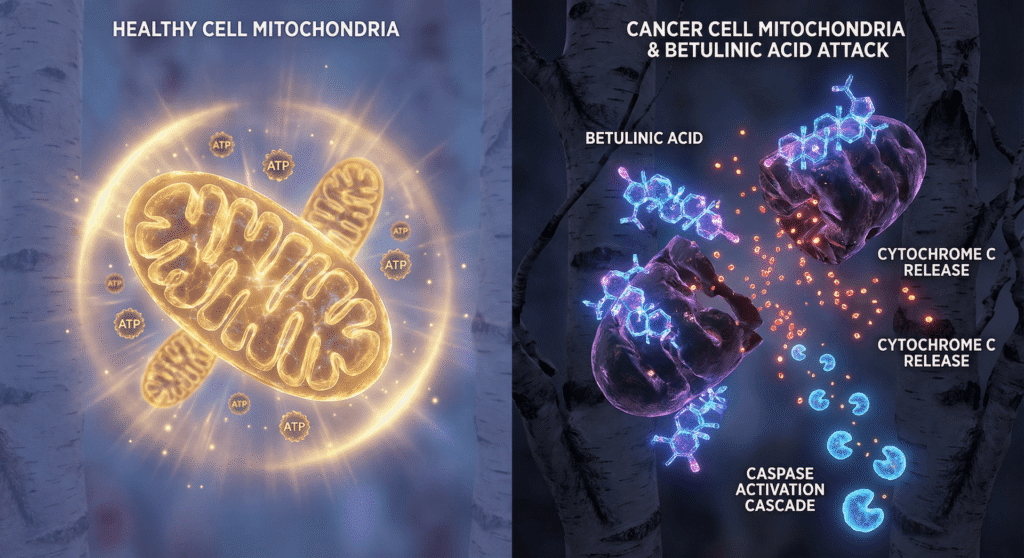
BA also generates reactive oxygen species (ROS) within cancer cells. While ROS in excess can damage cellular components, cancer cells are already operating under oxidative stress and are more vulnerable to additional ROS burden. BA exploits this vulnerability, pushing cancer cells past their breaking point while normal cells, with their superior antioxidant defenses, remain largely unaffected. This ROS generation activates pro-apoptotic signaling pathways, including the p38 and JNK mitogen-activated protein kinases, creating a perfect storm that leads to cancer cell death (Tan et al., 2003; Wang et al., 2019; Yi et al., 2021).
At the molecular level, BA modulates the expression of proteins in the Bcl-2 family, which regulate mitochondrial membrane permeability. It upregulates pro-apoptotic proteins like Bax while downregulating anti-apoptotic proteins like Bcl-2, tipping the balance decisively toward cell death. Additionally, BA has been shown to inhibit various cancer-promoting pathways, including NF-κB signaling, STAT3 activation, and Sp transcription factors, demonstrating its multi-targeted approach to cancer suppression (Chintharlapalli et al., 2007; Fulda, 2008; Mullauer et al., 2010; Rieber & Rieber, 2019; Wang et al., 2018).
The Cancer Types Most Susceptible to Betulinic Acid
Over the past three decades, hundreds of studies have investigated BA’s effects across a broad spectrum of cancer types. While nearly all tested cancers show some degree of sensitivity, certain malignancies have emerged as particularly responsive to BA treatment.
Melanoma remains the gold standard for BA research, with the most extensive body of evidence. In laboratory studies, BA demonstrates potent cytotoxicity against multiple melanoma cell lines (MEL-1, -2, -3, and -4) with IC₅₀ values (the concentration that kills 50% of cells) ranging from 0.5 to 4.8 µg/ml. More recent studies report IC₅₀ values between 2.21 and 15.94 µM across four different melanoma lines. The compound has advanced through preclinical development with support from the National Cancer Institute’s Rapid Access to Intervention Development (RAID) program, and clinical trials have been initiated to evaluate topical BA formulations for treating dysplastic nevi—precancerous lesions with potential to transform into melanoma (ClinicalTrials.gov, 2016; Kommera et al., 2024; Pisha et al., 1995; Rieber & Rieber, 2016; Sawant et al., 2022).
Glioblastoma: Crossing the Uncrossable Barrier
Glioblastoma multiforme is one of the most aggressive and lethal brain cancers, with a median survival of just 15 months despite aggressive treatment. One of the greatest challenges in treating brain cancers is the blood-brain barrier (BBB), a protective membrane that prevents most drugs from reaching the brain. Remarkably, BA can cross the BBB—a fantastic property that sets it apart from many potential therapeutic agents (Abranches et al., 2024; Marques et al., 2024).
Research demonstrates that BA effectively inhibits glioblastoma cell proliferation and induces apoptosis with IC₅₀ values ranging from 2 to 17 µg/mL. In preclinical studies, BA nanoparticles injected into mice with intracranial glioblastoma tumors crossed the BBB effectively and significantly prolonged survival. Moreover, BA has been shown to enhance the cytotoxic effects of temozolomide, the standard chemotherapy for glioblastoma, and suppress the aggressive, invasive behavior characteristic of these tumors. A particularly promising derivative called B10 (a glucose-conjugated BA) showed even greater efficacy, with doses of 25-50 mg/kg causing significant reductions in tumor weight and volume in mouse models (Abranches et al., 2024; Al-Ostoot et al., 2018; da Silva, 2017; Kommera et al., 2021; Parikh et al., 2024).
Neuroblastoma: Hope for Childhood Cancer
Neuroblastoma, a cancer that develops from immature nerve cells and primarily affects children, has shown remarkable sensitivity to BA. Studies demonstrate that BA induces apoptosis in neuroblastoma cells with an ED₅₀ (effective dose for 50% of cells) of 14-17 micrograms/ml. Importantly, BA works effectively against both N-MYC-positive neuroblastomas (which have a poor prognosis) and N-MYC-negative forms, and it has shown synergistic effects when combined with cisplatin, potentially allowing for lower doses of this toxic chemotherapy agent (Fulda et al., 1997; Oczeretko, 2022).
Breast Cancer: A Multifaceted Attack
Breast cancer research has revealed fascinating insights into BA’s mechanisms. BA not only induces apoptosis but also acts as a chemosensitizer, making resistant cancer cells more vulnerable to conventional therapies. The compound targets glucose-regulated protein 78 (GRP78), a stress response protein often overexpressed in breast cancers, triggering endoplasmic reticulum stress-mediated apoptosis. In preclinical studies using mice, daily doses of 10 mg/kg significantly reduced tumor growth and blocked metastasis to the lungs without observable toxicity. BA also reduces populations of myeloid-derived suppressor cells (MDSCs), immune cells that tumors recruit to evade immune destruction, thereby potentially enhancing the body’s natural anti-cancer immune response (Li et al., 2018; Qi et al., 2017).
Hematological Malignancies: Leukemia and Multiple Myeloma
BA has demonstrated significant activity against blood cancers. In multiple myeloma, a cancer of plasma cells, BA inhibits cell proliferation and induces apoptosis with effective concentrations of 5-25 µg/ml. Intriguingly, BA also inhibits autophagic flux—a cellular recycling process that myeloma cells exploit for survival under stress. In U937 human myeloid leukemia cells, BA induces cell cycle arrest at the G2/M phase and triggers apoptosis through ROS-dependent mechanisms. For Jurkat T-cell leukemia, the IC₅₀ at 24 hours is approximately 70 µmol/l (Rieber & Rieber, 2016; Yi et al., 2021; Zhao et al., 2012).
Solid Organ Cancers: Lung, Liver, Pancreas, and Beyond
BA’s anticancer portfolio extends impressively across solid organ cancers:
Lung cancer: IC₅₀ values range from 1.5 to 4.2 µg/mL, with BA demonstrating ability to inhibit cell proliferation, arrest cell cycle, and suppress metastatic potential. BA also shows synergistic effects with EGFR-tyrosine kinase inhibitors like gefitinib and osimertinib (Ames, 2024; Kommera et al., 2021; Ma et al., 2021).
Hepatocellular carcinoma (liver cancer): BA inhibits multiple liver cancer cell lines (HepG2, LM3, MHCC97H) in a time- and dose-dependent manner, inducing apoptosis through the mitochondrial pathway and significantly reducing pulmonary metastasis in animal models (Wang et al., 2018).
Pancreatic cancer: With an IC₅₀ of approximately 50 µM in Mia PaCa-2 and Panc-1 cells, BA not only kills pancreatic cancer cells but also targets cancer stem cells—the treatment-resistant cells often responsible for recurrence. BA activates AMPK signaling, inhibits epithelial-mesenchymal transition (a process that enables cancer spread), and enhances sensitivity to gemcitabine, the standard chemotherapy for pancreatic cancer (Fan et al., 2018).
Prostate cancer: BA induces apoptosis in both androgen-responsive (LNCaP) and androgen-refractory (DU145) prostate cancer cells at concentrations of 10-20 µM, working through stabilization of p53 and downregulation of NF-κB pathways. In mouse models, doses of 10 mg/kg effectively reduced tumor growth (Rieber & Rieber, 2016; Selzer et al., 2000).
Colorectal cancer: BA inhibits proliferation of colon cancer cells and xenograft tumor growth at concentrations of 5-10 µM, working through proteasome-dependent degradation of Sp transcription factors (Rieber & Rieber, 2016).
Ovarian cancer: With IC₅₀ values of 1.8-4.5 µg/mL, BA demonstrates time- and dose-dependent inhibition of ovarian cancer cell proliferation, induces apoptosis, and significantly impairs migration and invasion by suppressing epithelial-mesenchymal transition (Kommera et al., 2021; Li et al., 2020a; Li et al., 2020b).
Gastric cancer: BA inhibits gastric cancer cell growth with an IC₅₀ of 12.99 µg/ml in AGS cells, targeting cancer stem cells and inhibiting the tumor microenvironment through regulation of GRP78/TGF-β signaling and macrophage polarization (Rieber & Rieber, 2016; Zhang et al., 2023).
Cervical cancer: In HeLa cells, BA exhibits IC₅₀ values ranging from 30 to 66.75 µmol/l depending on treatment duration, inducing apoptosis through PI3K/Akt pathway inhibition and ROS-mediated endoplasmic reticulum stress (He et al., 2024; Xu et al., 2017).
Head and neck cancers: BA shows promise as both a direct anticancer agent and a radiosensitizer in squamous cell carcinomas of the head and neck (SCC9, SCC25 cell lines), potentially improving outcomes when combined with radiation therapy (Selzer et al., 2003; Thurnher et al., 2010).
Renal cell carcinoma: BA induces apoptosis in kidney cancer cells through modulation of the AKT/mTOR pathway in a time- and dose-dependent manner (Aydin et al., 2021; Aydin et al., 2022; Wang et al., 2018).
Human Dosing: Translating Laboratory Success to Clinical Reality
One of the most critical questions for any promising anticancer compound is: “What dose works in humans?” While BA has demonstrated impressive efficacy in cell cultures and animal models, translating these findings to human applications presents challenges.
Across the spectrum of cancer types, laboratory studies typically show effectiveness at IC₅₀ concentrations ranging from 1 to 50 µM or 1.8 to 17 µg/ml, depending on the specific cancer cell line and treatment duration (Kommera et al., 2021; Rieber & Rieber, 2016; Sawant et al., 2022). These concentrations provide a target for what blood or tissue levels might need to be achieved in patients.
Animal Studies: The Preclinical Bridge
Animal studies provide crucial dosing information that bridges laboratory and clinical research. The most commonly employed dose in mouse studies is 10 mg/kg per day, administered either intraperitoneally or orally. This dose consistently demonstrates tumor growth inhibition without significant toxicity across multiple cancer types including melanoma, breast cancer, and hepatocellular carcinoma (Qi et al., 2017; Rieber & Rieber, 2016).
Higher doses have also been explored:
- 20-67 mg/kg for anti-inflammatory effects (de Carvalho et al., 2022; Martins et al., 2022)
- 25-50 mg/kg for B10 (a BA derivative) in glioma models (da Silva, 2017)
- 30 mg/kg in various cancer models (Kommera et al., 2021)
- Up to 100 mg/kg showing no toxicity in safety studies (Mullauer et al., 2010; Zhao et al., 2012)
- Even 500 mg/kg has been mentioned as safe in some reports (Al-Ghadban et al., 2023)
It’s important to note that doses in mice don’t translate directly to humans due to differences in metabolism and body surface area. Using standard conversion factors, a 10 mg/kg dose in mice would approximate to roughly 0.8 mg/kg in humans (or about 56 mg for a 70 kg person), though this is a rough estimate requiring careful clinical validation (Nair & Jacob, 2016).
Clinical Trials: The Human Evidence
While BA shows tremendous promise in preclinical studies, human clinical trials remain limited, representing a significant gap in our knowledge:
Topical melanoma trial (NCT00346502): A phase I/II clinical trial evaluated 20% betulinic acid ointment for treating dysplastic nevi. The study planned daily application for 4 weeks followed by surgical removal and histological comparison with untreated lesions. Unfortunately, this trial was suspended in 2013 due to funding issues, and results were never published (ClinicalTrials.gov, 2016; Kommera et al., 2024).
Cutaneous metastatic melanoma (NCT00701987): This phase 1 pilot trial with 12 participants evaluated topical ALS-357 (betulinic acid formulation) for safety, tolerability, and efficacy. The drug was applied in ointment form for 4 weeks in four different frequency groups (twice weekly, every other day, daily, or twice daily), with the TUNEL assay used to evaluate apoptosis in biopsies. Limited information about outcomes is publicly available (ClinicalTrials.gov, 2016; DrugBank, 2022).
Bevirimat (BA derivative): In HIV treatment trials, this 3′-O-(3′,3′-dimethylsuccinyl) betulinic acid derivative was tested at oral doses of 150-250 mg, showing dose-dependent activity and good tolerability. While these were HIV trials, not cancer trials, they provide valuable pharmacokinetic data demonstrating that BA derivatives can achieve therapeutic blood levels when administered orally to humans with acceptable safety profiles (Smith et al., 2007).
One of the most significant obstacles limiting BA’s clinical development is its poor oral bioavailability. Like many natural compounds with beneficial properties, BA is highly lipophilic (fat-soluble) with limited water solubility, which restricts absorption from the gastrointestinal tract (Al-Ghadban et al., 2023; Das et al., 2014; Das et al., 2025).
Standard BA formulations show oral bioavailability of less than 10%. However, pharmaceutical scientists have developed several innovative solutions (Al-Ghadban et al., 2023):
Spray-dried mucoadhesive formulations: These show 3.9-fold increases in maximum blood concentration (Cmax) and 7.41-fold increases in area under the curve (AUC, a measure of total drug exposure), with significant improvements in anticancer effects in lung cancer models (Das et al., 2014).
Self-nanoemulsifying drug delivery systems (SNEDDS): Using fish oil as a carrier, these formulations achieved 16.66 times greater bioavailability compared to free BA in rat studies (Alshbool et al., 2024).
Nanoparticle formulations: Various nanoparticle systems including PLGA nanoparticles, liposomes, and protein-based carriers have demonstrated markedly improved bioavailability, extended circulation half-life, and enhanced tumor accumulation (Parikh et al., 2024; Sharma et al., 2018).
These formulation advances are critical for translating BA’s laboratory promise into clinical reality.
Safety Profile: The Selective Advantage
Perhaps one of BA’s most attractive features is its remarkable safety profile. Unlike conventional chemotherapy, which often causes severe systemic toxicity, BA demonstrates selective cytotoxicity—it targets cancer cells while largely sparing normal tissues (Fulda, 2008; Mullauer et al., 2010).
- Normal melanocytes remain relatively resistant to BA compared to melanoma cells (Zhao et al., 2012)
- Peripheral blood lymphocytes and human skin fibroblasts show high resistance to BA treatment (Mullauer et al., 2010)
- In cancer/normal cell co-culture studies, BA selectively killed cancer cells while sparing normal fibroblasts (Das et al., 2015)
- Animal studies show complete lack of toxicity at doses up to 100 mg/kg body weight (Mullauer et al., 2010; Zhao et al., 2012)
- Some studies report safety at even 500 mg/kg (Al-Ghadban et al., 2023)
- Histological examination of organs (liver, kidney, spleen, lung) in BA-treated animals shows normal tissue architecture with no signs of toxicity (Qi et al., 2017)
On human normal dermatoblast cell lines, BA was one-half to one-fifth as toxic as doxorubicin (a standard chemotherapy drug), providing a substantial therapeutic window (Wikipedia, 2024).
Why does BA preferentially kill cancer cells? Several factors contribute:
- Metabolic differences: Cancer cells typically have altered mitochondrial function and higher metabolic rates, making them more vulnerable to mitochondrial disruption (Fulda & Kroemer, 2009).
- Oxidative stress: Cancer cells operate under higher baseline oxidative stress, leaving them more susceptible to ROS-generating agents like BA (Tan et al., 2003; Yi et al., 2021).
- Survival pathway dependencies: Many cancers become dependent on specific survival pathways (like NF-κB, STAT3, or Sp transcription factors) that BA inhibits, while normal cells have functional redundancy (Chintharlapalli et al., 2007; Rieber & Rieber, 2019; Rieber & Rieber, 2016).
Limited human data from clinical trials and case studies suggest good tolerability:
- Topical BA formulations showed no significant adverse events in melanoma trials (Kommera et al., 2024)
- Oral bevirimat (BA derivative) at doses up to 250 mg was well tolerated in HIV patients (Smith et al., 2007)
- A study in dogs showed BA was well tolerated at 8× recommended dose with no adverse reactions except minor effects attributable to pre-existing conditions (ClinicalTrials.gov, 2019)
Combination Therapies: Strength in Numbers
While BA shows impressive standalone activity, its true potential may lie in combination approaches. Research increasingly demonstrates that BA can enhance the efficacy of conventional cancer treatments while potentially reducing required doses and associated toxicities.
Gemcitabine + BA: In pancreatic cancer, BA enhanced gemcitabine’s effects and reversed cancer stem cell properties induced by chemotherapy (Fan et al., 2018).
Taxanes + BA: Combined treatment of melanoma cells with BA and either paclitaxel or docetaxel generated additive to synergistic interactions. BA similarly enhances taxol chemosensitivity in breast cancer through apoptosis induction and G2/M cell cycle arrest (Li et al., 2018; Sawant et al., 2022).
Cisplatin + BA: In neuroblastoma, BA-cisplatin combinations showed synergistic activity on cell growth inhibition and apoptotic cell death (Abranches et al., 2024; Oczeretko, 2022).
Temozolomide + BA: BA enhances the cytotoxic effects of temozolomide in glioblastoma, the standard chemotherapy for this aggressive brain cancer (Abranches et al., 2024).
EGFR-TKIs + BA: In lung cancer with wild-type EGFR, BA synergizes with gefitinib and osimertinib to overcome intrinsic resistance, activating autophagy and inducing cell cycle arrest (Ames, 2024).
BA has demonstrated radiosensitizing properties in several cancers:
- In melanoma cells, BA combined with irradiation showed additive effects on growth inhibition (Selzer et al., 2000)
- In head and neck squamous cell carcinomas, BA potentiated the effects of radiation, suggesting potential as a radiosensitizing agent for radioresistant tumors (Thurnher et al., 2010)
- The compound appears to work by modulating DNA damage response pathways and inhibiting repair mechanisms that cancer cells employ to survive radiation (Abranches et al., 2024)
Emerging evidence suggests BA may enhance anti-tumor immunity:
- BA reduces populations of myeloid-derived suppressor cells (MDSCs), immune cells that tumors recruit to suppress anti-cancer immune responses (Qi et al., 2017)
- The compound modulates macrophage polarization, shifting the tumor microenvironment from immunosuppressive toward immune-activating (Zhang et al., 2023)
- These immune-modulating effects suggest potential synergy with checkpoint inhibitors and other immunotherapies, though this remains to be formally tested
From Bench to Bedside: Challenges and Future Directions
Despite three decades of research and promising preclinical data, BA has not yet achieved widespread clinical use. Several challenges must be addressed:
Poor water solubility and low oral bioavailability remain the primary obstacles. While advanced formulations show promise, none have achieved regulatory approval. Future directions include (Das et al., 2025; Das et al., 2014):
- Continued development of nanoparticle delivery systems
- Structural modifications to improve pharmaceutical properties
- Prodrug strategies that convert to active BA within tumors
- Novel administration routes (intravenous, intraperitoneal, intrathecal for brain cancers)
Challenge 2: Limited Clinical Data
The scarcity of completed human clinical trials represents a critical knowledge gap. Reasons for limited clinical development include:
- Funding challenges for natural products that may be difficult to patent
- Focus of pharmaceutical industry on synthetic compounds
- Formulation difficulties that made early clinical development challenging
- Suspension of initial trials before completion
Addressing this requires:
- Renewed clinical trial efforts with improved formulations
- Public-private partnerships to fund natural product research
- Combination trials with approved therapies
- Careful pharmacokinetic studies to establish optimal human dosing
Challenge 3: Source and Synthesis
While birch bark is abundant, extracting sufficient BA for large-scale pharmaceutical use presents sustainability concerns. Alternative approaches include (Kommera et al., 2024):
- Chemical synthesis from betulin (a related compound more abundant in birch bark)
- Biotransformation using engineered microorganisms
- Plant cell culture systems
- Total chemical synthesis, though this remains complex and expensive
Challenge 4: Derivative Development
While BA shows promise, derivatives might offer improved properties. Hundreds of BA analogues have been synthesized and tested, with some showing superior activity. The B10 derivative (glucose-conjugated BA) demonstrates particularly promising results in glioblastoma. Future medicinal chemistry efforts should focus on (da Silva, 2017; Parikh et al., 2024; Pluta et al., 2012; Zhao et al., 2024):
- Improving target specificity
- Enhancing blood-brain barrier penetration
- Optimizing pharmacokinetic properties
- Maintaining or improving safety profiles
The Path Forward: Promise and Perseverance
Betulinic acid represents a compelling example of nature’s pharmacological potential. Its selective toxicity toward cancer cells, multi-targeted mechanism of action, impressive safety profile, and activity across a broad spectrum of malignancies distinguish it from many conventional therapies. The compound’s ability to cross the blood-brain barrier makes it particularly attractive for notoriously difficult-to-treat brain cancers, while its chemosensitizing properties suggest valuable roles in combination regimens.
The research is clear: BA works. It kills cancer cells in petri dishes. It shrinks tumors in mice. It does so with minimal toxicity. The question is no longer whether BA has anticancer properties—decades of research have firmly established that—but rather how to effectively deliver it to human patients at concentrations that achieve therapeutic benefit.
For cancers with the most robust evidence such as melanoma, glioblastoma, neuroblastoma, and breast cancer, it is obvious that BA warrants serious consideration for clinical development and utilization. The convergence of three factors may finally enable this translation: (1) advanced nanoformulation technologies that overcome bioavailability limitations, (2) renewed interest in natural products as cancer therapy leads, and (3) the growing recognition that selective, multi-targeted agents may offer advantages over highly specific but readily circumvented therapies.
The journey from laboratory bench to patient bedside is long, expensive, and uncertain. BA has traveled much of this path but not yet reached the destination. With continued research investment, innovative formulation strategies, and commitment to rigorous clinical testing, this remarkable natural compound may yet fulfill its promise as a valuable weapon in humanity’s ongoing battle against cancer.
For patients and advocates following BA’s development, the message is one of cautious optimism. The science is solid, the potential is real, but clinical validation remains incomplete. As with many promising natural compounds, the challenge lies not in demonstrating activity but in navigating the complex path to regulatory approval and clinical implementation.
I personally love to use BA in my practice because it has such an excellent risk-to-benefit ratio. The benefits far outweigh the risk. However, this does not mean that I would rely on this alone for treatment for the many reasons stated above. I have recently seen excellent benefit using Chaga (sourced from birch bark) combined with MCS formulas’ liposomal betulinic acid. Because dosing has not really been fully established and ranges from around 10 mg-100 mg/kg (and could go as high as 500 mg/kg), which means for the average 150 lb person, this translates to approximately 680 mg-6,800 mg (34,000 mg in the highest!) as a daily dose. However, liposomal formulations show much better absorption and thus can be up to 16× less of an oral dose. Thus, for MCS formulas’ liposomal form, I shoot for the lowest dose being 1 capsule (50 mg) twice per day but can easily go up to 4 capsules (200 mg) twice per day in high-need cases and larger individuals. Just to emphasize, this is all based on my experience, and the dosing is based on theory extracted from the scientific literature.
The story of betulinic acid and cancer is far from over. It may, in fact, be just beginning.
Author’s Note
This article is intended for educational purposes and represents a comprehensive review of published scientific literature on betulinic acid and cancer. It should not be construed as medical advice. Betulinic acid is not approved for cancer treatment, and individuals should consult qualified healthcare professionals regarding cancer prevention and treatment options. While the scientific evidence for betulinic acid’s anticancer properties is substantial, clinical validation in humans remains limited, and the compound should only be used under appropriate medical supervision in approved clinical trials.
Abranches, M., Silva, J., & Ferreira, H. (2024). Betulinic Acid for Glioblastoma Treatment: Reality, Challenges and Future Perspectives.
Al-Ghadban, S., Al-Qassas, R., & Al-Taani, B. (2023). A Novel Betulinic Acid Analogue: Synthesis, Solubility, Antitumor Activity, and Molecular Docking Studies. Pharmaceuticals, 16(8), 1083.
Ali-Seyed, M., Jantan, I., Vijayaraghavan, K., & Bukhari, S. N. A. (2016). Betulinic acid: Recent advances in chemical modifications, effective delivery, and molecular mechanisms of a promising anticancer therapy. Chemical Biology & Drug Design, 87(4), 517–536.
Al-Ostoot, F. H., Al-attar, A., & Al-Tannak, N. F. (2018). DDIS-23. BETULINIC ACID SUPPRESS GLIOBLASTOMA CELLS PROLIFERATION THROUGH ER STRESS APOPTOTIC PATHWAY.
Alshbool, F. Z., Al-Ani, I. H., & Al-Qaisi, Z. (2024). Enhanced Bioavailability of Betulinic Acid Through the Utilization of a Self-Nanoemulsifying Drug Delivery System (SNEDDS) Formulated with Fish Oil. SSRN.
Ames, B. N. (2024). Combination of betulinic acid and EGFR-TKIs exerts synergistic anti-proliferative effect on wild-type EGFR lung cancer cells via activating autophagy and inducing cell cycle arrest. Naunyn-Schmiedeberg’s Archives of Pharmacology.
Aydin, S., Ciftci, O., & Cetin, A. (2021). Effects of betulinic acid on AKT/mTOR pathway in renal cell carcinoma. Urology Research and Practice, 47(4), 221–226.
Aydin, S., Ciftci, O., & Cetin, A. (2022). Betulinic acid may modulate autophagy in renal cell carcinoma cells. Urologia Internationalis, 107(1), 74–81.
Chintharlapalli, S., Papineni, S., Ramaiah, S. K., & Safe, S. (2007). Betulinic acid inhibits prostate cancer growth through inhibition of specificity protein transcription factors. Cancer Research, 67(6), 2816–2823.
ClinicalTrials.gov. (2016). Evaluation of 20% Betulinic Acid Ointment for Treatment of Dysplastic Nevi. U.S. National Library of Medicine.
ClinicalTrials.gov. (2019). Safety, Tolerability and Behavioural Effects of Souroubea-Platanus Based Formulation in Dogs. U.S. National Library of Medicine.
da Silva, G. (2017). Betulinic acid derivative B10 inhibits glioma cell proliferation through cell cycle arrest and apoptosis. Biomedicine & Pharmacotherapy, 95, 1845–1853.
Das, S., Das, S., & Basu, S. P. (2014). Approaches to improve the oral bioavailability and effects of novel anticancer molecules and natural compounds on novel anticancer molecules. Journal of Controlled Release, 196, 327–338.
Das, S., Kumar, M., & Verma, M. (2015). A potent tumoricidal co-drug ‘Bet-CA’—An ester derivative of betulinic acid and chlorambucil. Scientific Reports, 5(1), 7762.
Das, S., Jana, S., & Dash, S. (2025). Advances and challenges in betulinic acid therapeutics and delivery systems. RSC Advances, 15(46), 29775–29801.
de Carvalho, A. P. A., Oliveira, J. P. F., & de Almeida, J. R. G. S. (2022). Anti-Inflammatory Activities of Betulinic Acid: A Review. Frontiers in Pharmacology, 13.
DrugBank. (2022). Betulinic Acid Phase 1 Trials for Melanoma Treatment.
Fan, C., Chen, J., Wang, Y., & Wong, Y. (2018). Betulinic acid inhibits stemness and EMT of pancreatic cancer cells by interfering with STAT3 and FAK signaling. OncoTargets and Therapy, 11, 7175–7185.
Fulda, S. (2008). Betulinic Acid for Cancer Treatment and Prevention. International Journal of Molecular Sciences, 9(6), 1096–1107.
Fulda, S., & Kroemer, G. (2009). Targeting mitochondrial apoptosis by betulinic acid in human cancers. Drug Discovery Today, 14(17–18), 885–890.
Fulda, S., Friesen, C., Los, M., Scaffidi, C., Mier, W., Benedict, M., Nunez, G., Krammer, P. H., Peter, M. E., & Debatin, K. M. (1997). Betulinic acid induces apoptosis in human neuroblastoma cell lines. Medical and Pediatric Oncology, 29(5), 379–380.
He, Q., Wang, Y., & Zhang, Q. (2024). Betulinic acid induces apoptosis of HeLa cells via ROS-dependent endoplasmic reticulum stress and mitochondrial dysfunction pathway. Journal of Gynecologic Oncology, 35(4), e50.
Kommera, H., Behera, A. K., & Kesharwani, P. (2021). Anticancer Potential of Betulonic Acid Derivatives. Anti-Cancer Agents in Medicinal Chemistry, 21(14), 1836–1845.
Kommera, H., Singh, S., & Behera, A. K. (2024). Chemopreventive and Chemotherapeutic Potential of Betulin and Its Derivatives: Mechanistic Insights From In Vitro, In Vivo and In Silico Studies. Food Science & Nutrition, 12(6), 4057–4074.
Li, J., He, Y., & Weng, Y. (2018). Betulinic acid chemosensitizes breast cancer by triggering ER stress and apoptosis and reducing the population of myeloid-derived suppressor cells. Cell Death & Disease, 9(6), 598.
Li, L., Liu, C., Xie, X., & Zhou, J. (2020a). Betulinic acid induces apoptosis and impairs migration and invasion in a mouse model of ovarian cancer. Journal of Food Biochemistry, 44(7), e13278.
Li, L., Liu, C., & Zhou, J. (2020b). Betulinic acid induces apoptosis and impairs migration and invasion in a mouse model of ovarian cancer. Journal of Food Biochemistry, 44(7).
Ma, J., Zhou, Y., & Li, M. (2021). Lung Cancer Inhibition by Betulinic Acid Nanoparticles via ROS-Mediated Apoptosis and STAT3/MMP-2 Pathway. Frontiers in Oncology, 11.
Marques, S., da Silva, A. B., & Ayres-Silva, A. (2024). Betulinic Acid for Glioblastoma Treatment. International Journal of Molecular Sciences, 25(4), 2182.
Martins, G., da Silva, L., & de Souza, E. (2022). Anti-Inflammatory Activities of Betulinic Acid: A Review. Molecules, 27(10), 3290.
Mullauer, F. B., Kessler, J. H., & Medema, J. P. (2010). Betulinic acid, a natural compound with potent anticancer effects. Anti-Cancer Drugs, 21(3), 215–227.
Nair, A. B., & Jacob, S. (2016). A simple practice guide for dose conversion between animals and human. Journal of Basic and Clinical Pharmacy, 7(2), 27–31.
Oczeretko, A. (2022). Betulinic Acid and Cisplatin Inhibit Metastasis Related Genes in Neuroblastoma Cells. Central European Journal of Urology, 75(1), 108–115.
Parikh, A., Patel, K., & Patel, K. (2024). Advancements in Betulinic Acid-Loaded Nanoformulations for Cancer Therapy. Journal of Pharmaceutical Sciences.
Pezzuto, J. M. (1995). Discovery of betulinic acid as a selective inhibitor of human melanoma that functions by induction of apoptosis. Nature Medicine, 1(10), 1046–1051.
Pisha, E., Chai, H., Lee, I.-S., Chagwedera, T. E., Farnsworth, N. R., Cordell, G. A., Beecher, C. W. W., Fong, H. H. S., Kinghorn, A. D., Brown, D. M., Wani, M. C., Wall, M. E., Hieken, T. J., Das Gupta, T. K., & Pezzuto, J. M. (1995). Discovery of betulinic acid as a selective inhibitor of human melanoma that functions by induction of apoptosis. Nature Medicine, 1(10), 1046–1051.
Pluta, R., Pluta, A., & Ułamek-Kozioł, M. (2012). New ionic derivatives of betulinic acid as highly potent anti-cancer agents. Bioorganic & Medicinal Chemistry Letters, 22(4), 1643–1647.
Qi, Y., Zhang, F., & Li, Z. (2017). Betulinic acid impairs metastasis and reduces immunosuppressive cells in breast cancer models. Oncotarget, 8(70), 115664–115675.
Rieber, M., & Rieber, M. S. (2016). Betulinic acid and the pharmacological effects of tumor suppression. Molecular Medicine Reports, 14(6), 5461–5466.
Rieber, M., & Rieber, M. S. (2019). Betulinic Acid Induces ROS-Dependent Apoptosis and S-Phase Arrest in Ovarian Cancer Cells. Oxidative Medicine and Cellular Longevity, 2019, 1–11.
Sawant, S. S., Kshirsagar, S. J., & Patil, S. V. (2022). Additive Interactions between Betulinic Acid and Two Taxanes in In-Vitro Models of Human Melanoma. Current Drug Discovery Technologies, 19(3), 56–62.
Selzer, E., Pimentel, E., Wacheck, V., Schlegel, W., Pehamberger, H., Jansen, B., & Kodym, R. (2000). Effects of betulinic acid alone and in combination with irradiation in human melanoma cells. Journal of Investigative Dermatology, 114(5), 935–940.
Selzer, E., Thurnher, D., & Formanek, M. (2003). Betulinic acid: a new cytotoxic compound against malignant head and neck cancer cells. The Laryngoscope, 113(8), 1386–1391.
Sharma, G., Sharma, A. R., & Nam, J.-S. (2018). Therapeutic applications of betulinic acid nanoformulations. Molecules, 23(1), 191.
Smith, P. F., Ogundele, A., & Forrest, A. (2007). Phase I and II study of bevirimat, a novel inhibitor of human immunodeficiency virus type 1 maturation, in treatment-experienced subjects. Antimicrobial Agents and Chemotherapy, 51(10), 3574–3581.
Tan, Y., Yu, R., & Pezzuto, J. M. (2003). Betulinic acid-induced programmed cell death in human melanoma cells involves mitogen-activated protein kinase activation. Clinical Cancer Research, 9(7), 2866–2875.
Thurnher, D., Knerer, B., & Formanek, M. (2010). Betulinic acid a radiosensitizer in head and neck squamous cell carcinoma. Head & Neck, 32(3), 391–398.
Wang, H., Dong, X., Liu, Z., Zhu, S., Liu, H., Fan, W., Li, L., & Guo, Y. (2018). Betulinic acid induces apoptosis and suppresses metastasis in hepatocellular carcinoma cell lines in vitro and in vivo. Journal of Cellular and Molecular Medicine, 22(11), 5431–5440.
Wang, L., Zhang, J., & Li, R. (2019). Betulinic acid induces apoptosis and inhibits metastasis of human choroidal melanoma. OncoTargets and Therapy, 12, 2949–2958.
Wikipedia. (2024). Betulinic acid. Wikimedia Foundation.
Xu, T., Pang, Q., Wang, Y., Yan, X., & Liu, C. (2017). Betulinic acid induces apoptosis by regulating PI3K/Akt signaling and mitochondrial pathways in human cervical cancer cells. International Journal of Molecular Medicine, 40(5), 1438–1444.
Yi, E., Oh, S., & Lee, S. (2021). The anti-cancer effect of betulinic acid in U937 human leukemia cells through generation of reactive oxygen species. Cancers, 13(8), 1937.
Zhang, H., Wang, Z., & Qu, X. (2023). Betulinic Acid Inhibits the Stemness of Gastric Cancer Cells by Regulating GRP78/TGF-β Signaling to Polarize Tumor-Associated Macrophages. Cancers, 15(4), 1184.
Zhao, G., Zhao, L., & Guo, Y. (2012). Betulinic acid inhibits autophagic flux and induces apoptosis in multiple myeloma cells. Acta Pharmacologica Sinica, 33(11), 1427–1434.
Zhao, L., Li, C., & Zhang, Y. (2024). Novel C-3 and C-20 derived analogs of betulinic acid as potent anticancer agents. RSC Advances.

